Unlocking the Enigmatic World of Stereochemistry and Organic Reactions: A Comprehensive Guide

4.1 out of 5
| Language | : | English |
| File size | : | 97377 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 640 pages |
| Hardcover | : | 440 pages |
| Item Weight | : | 1.6 pounds |
| Dimensions | : | 6.14 x 0.94 x 9.21 inches |
to Stereochemistry
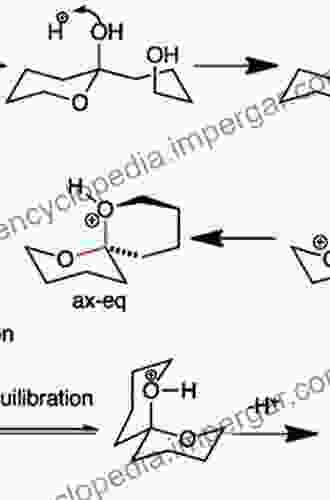
Stereochemistry, a captivating field within chemistry, delves into the spatial arrangement of atoms and groups within molecules, offering profound insights into how their three-dimensional structures influence their properties and reactivity. This intricate dance of molecular architecture unveils the fundamental differences between molecules that may share the same molecular formula but exhibit distinct stereochemical configurations.
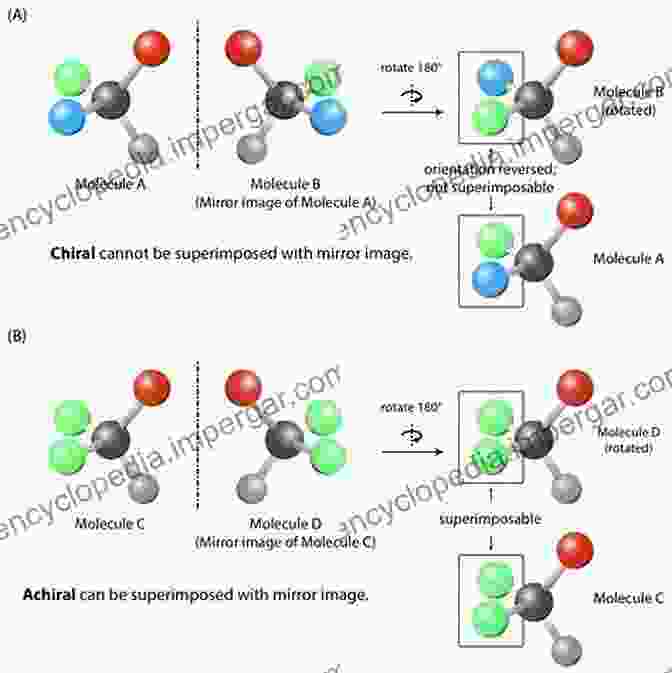
Chirality, a hallmark of stereochemistry, arises from the absence of a plane of symmetry within a molecule. These chiral molecules exist in two mirror-image forms, akin to our left and right hands, known as enantiomers. While indistinguishable in terms of their physical properties, enantiomers exhibit fascinating differences in their interactions with other chiral entities, be it biological molecules or chiral catalysts.
Stereochemistry and Organic Reactions
The significance of stereochemistry extends beyond the realm of molecular recognition and finds profound applications in organic reactions. Stereochemistry dictates the course of reactions, influences the regio- and stereoselectivity of outcomes, and ultimately governs the synthesis of target molecules with desired properties.
Stereoselectivity, a crucial aspect of organic reactions, refers to the preferential formation of one stereoisomer over others. This exquisite control over molecular architecture is essential for synthesizing complex natural products, pharmaceuticals, and functional materials with specific biological activities and desired properties.
Tools for Stereochemical Control
Chemists have developed an arsenal of strategies to control stereochemistry in organic reactions. These tactics include:
- Chiral Auxiliaries: These temporary molecular attachments bias the reaction towards the formation of a specific stereoisomer.
- Chiral Catalysts: These specialized catalysts facilitate reactions with high enantio- or diastereoselectivity.
- Asymmetric Synthesis: This powerful approach utilizes chiral reagents or catalysts to construct chiral molecules with high enantiomeric purity.
Applications of Stereochemistry in Drug Discovery and Development
Stereochemistry plays a paramount role in drug discovery and development, where the biological activity and efficacy of pharmaceuticals can be profoundly influenced by their stereochemical configurations. Enantiomers of the same drug molecule may exhibit vastly different pharmacological properties, pharmacokinetic profiles, and side effect profiles.
Understanding stereochemistry is critical for optimizing drug potency, selectivity, and reducing adverse effects. It also guides the design of new drugs with improved efficacy and reduced toxicity.
Stereochemistry, with its intricate interplay of spatial arrangements and molecular properties, offers a profound understanding of the three-dimensional world of molecules. Its significance in organic reactions empowers chemists to control and predict the outcomes of reactions, unlocking the potential for the synthesis of complex and valuable molecules. As our knowledge of stereochemistry continues to expand, it will undoubtedly shape the future of chemistry and fuel groundbreaking discoveries in various scientific disciplines.
4.1 out of 5
| Language | : | English |
| File size | : | 97377 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 640 pages |
| Hardcover | : | 440 pages |
| Item Weight | : | 1.6 pounds |
| Dimensions | : | 6.14 x 0.94 x 9.21 inches |
Do you want to contribute by writing guest posts on this blog?
Please contact us and send us a resume of previous articles that you have written.
 Book
Book Novel
Novel Page
Page Chapter
Chapter Text
Text Story
Story Genre
Genre Reader
Reader Library
Library Paperback
Paperback E-book
E-book Magazine
Magazine Newspaper
Newspaper Paragraph
Paragraph Sentence
Sentence Bookmark
Bookmark Shelf
Shelf Glossary
Glossary Bibliography
Bibliography Foreword
Foreword Preface
Preface Synopsis
Synopsis Annotation
Annotation Footnote
Footnote Manuscript
Manuscript Scroll
Scroll Codex
Codex Tome
Tome Bestseller
Bestseller Classics
Classics Library card
Library card Narrative
Narrative Biography
Biography Autobiography
Autobiography Memoir
Memoir Reference
Reference Encyclopedia
Encyclopedia C F A Culling
C F A Culling Philippe Gutton
Philippe Gutton Robert Jervis
Robert Jervis Ann Embry
Ann Embry Candi Byrne
Candi Byrne Michael E Mccullough
Michael E Mccullough Jeremy Enfinger
Jeremy Enfinger Chuck Solomon
Chuck Solomon John Nores
John Nores Jeff Benedict
Jeff Benedict Miriam Beloglovsky
Miriam Beloglovsky Baojiang Sun
Baojiang Sun Sikander Sultan
Sikander Sultan Werner Herzog
Werner Herzog Danielle Stringer
Danielle Stringer Jennifer Allwood
Jennifer Allwood Theodore Dwight Bozeman
Theodore Dwight Bozeman Ron Levy
Ron Levy Wedding Mentor
Wedding Mentor P Parvatha Reddy
P Parvatha Reddy
Light bulbAdvertise smarter! Our strategic ad space ensures maximum exposure. Reserve your spot today!

 W. Somerset MaughamBrain-Friendly Strategies for Unlocking Your Child's Learning Potential
W. Somerset MaughamBrain-Friendly Strategies for Unlocking Your Child's Learning Potential Isaac AsimovFollow ·16.6k
Isaac AsimovFollow ·16.6k Fredrick CoxFollow ·17.7k
Fredrick CoxFollow ·17.7k Ismael HayesFollow ·5k
Ismael HayesFollow ·5k Pablo NerudaFollow ·6.8k
Pablo NerudaFollow ·6.8k Jeffrey HayesFollow ·5.9k
Jeffrey HayesFollow ·5.9k Richard AdamsFollow ·8.3k
Richard AdamsFollow ·8.3k Cole PowellFollow ·14.5k
Cole PowellFollow ·14.5k Ben HayesFollow ·15.4k
Ben HayesFollow ·15.4k

 Terence Nelson
Terence NelsonSocial Dynamics in Systems Perspective: New Economic...
The world we live in is a complex and...

 Deacon Bell
Deacon BellUnlock the Secrets of Treasury Process Internal Controls:...
In today's competitive business...

 Finn Cox
Finn CoxThe Path Ahead: Green Energy and Technology
Embark on the...

 Rob Foster
Rob FosterThermodynamics of Surfaces and Capillary Systems: A...
Surfaces and...

 Nathan Reed
Nathan ReedUnlock the Secrets to Writing Remarkable Business School...
Embarking on the journey to business...

 David Foster Wallace
David Foster WallacePrinciples and Applications, Second Edition: Your Gateway...
In the ever-evolving realm of...
4.1 out of 5
| Language | : | English |
| File size | : | 97377 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 640 pages |
| Hardcover | : | 440 pages |
| Item Weight | : | 1.6 pounds |
| Dimensions | : | 6.14 x 0.94 x 9.21 inches |